“Reading World on the Edge brings topics ranging from crop failures to state failures together in a way that no series of blog posts could…Brown draws these connections with both scientific rigor and the patience of a committed teacher.” —Jeff McIntire-Strasburg, Sustainablog.org
Chapter 6. Designing a New Materials Economy: Materials and the Environment
The materials used in our modern economy fall into three categories. The first is metals, including steel, aluminum, copper, zinc, and lead. The second category is nonmetallic minerals, such as stone, sand, gravel, limestone, and clay—materials that are used directly in the building of highways, roads, and buildings or in manufacturing concrete. This group also includes three minerals—phosphate, potash, and lime—that are used in agriculture to raise soil fertility. (See Table 6-2.) The final group of raw materials includes those of organic origin, such as wood from the forest sector and cotton, wool, and leather from agriculture.12
In the nonmetallic category, stone at 11 billion tons produced per year and sand and gravel at 9 billion tons a year totally dominate other minerals. But stone, sand, and gravel are usually available locally and do not involve long-distance transport. Used primarily for the construction of roads, parking lots, and buildings, these materials are chemically inert. Once stone or gravel is in place in a roadbed, it may last for generations or even centuries.13
This chapter concentrates on metals because their mining and processing are so environmentally destructive and energy-intensive. Their production uses seemingly endless quantities of energy to remove earth to reach the ore, extract it, transport it to a smelter, and then process it into a pure metal. What's more, much of this energy comes from coal, which itself must be mined. Over time, as high-grade ores have been depleted, miners have shifted to lower-grade ores, inflicting progressively more environmental damage with each ton of metal produced.14
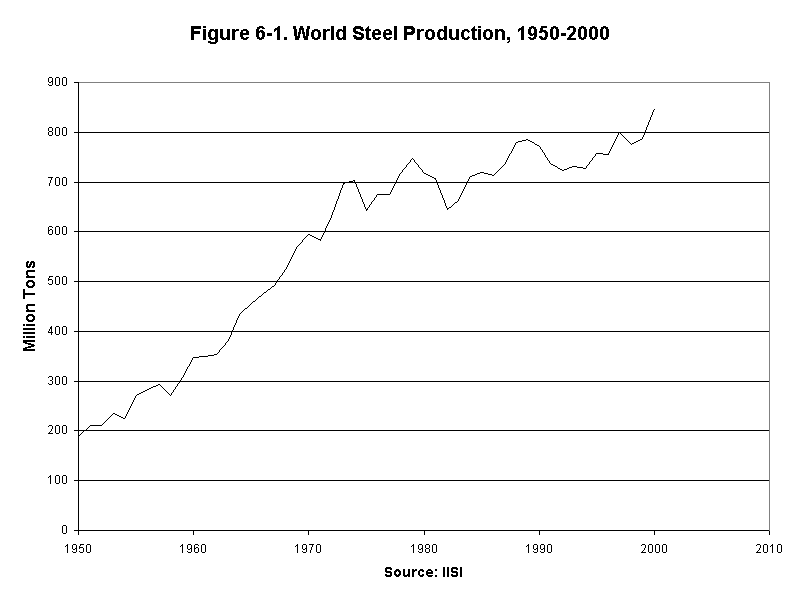
Ever since the Industrial Age began, steel production has been a basic indicator of industrialization and economic modernization. In the late twentieth century, the Soviet Union was the international steel giant. In the early 1990s, however, the breakdown of Soviet steel output paralleled the breakdown of the Soviet regime. Currently, China leads the world in steel production, followed by the United States and Japan. In quantity, the 833 million tons of raw steel produced each year (see Figure 6-1) dwarfs the use of all other metals combined. It compares with 24 million tons of aluminum and 13 million tons of copper, the second and third ranking metals. While steel consists predominantly of iron, it is an alloy, and many of its attractive characteristics come from the addition of small quantities of other metals such as zinc, magnesium, and nickel.15
World steel production per person reached its historical high in 1979 and has since dropped by 20 percent. The decline reflects a shift to smaller cars, the partial collapse of the former Soviet economy, and a shift in the growth of advanced industrial economies from heavy industry to services, especially information services.16
Every year, 1.4 billion tons of ore are mined worldwide to produce steel primarily for automobiles, household appliances, and construction. A comparable quantity of ore is mined to produce 13 million tons of copper. In an age when open pit mining has largely replaced underground mines, vast areas are physically disfigured. The mine tailings are then left behind—often disrupting the flow of nearby streams and contaminating water supplies. Anything that reduces the use of steel, particularly that produced from virgin ore, markedly lightens the human footprint on the earth.17
Although aluminum production is quite small compared with steel, the 24 million tons produced annually greatly understate aluminum's role because it is such a light, low-density metal. Australia produces one third of the world's aluminum-containing bauxite, with Guinea, Jamaica, and Brazil also contributing significantly to the world total.18
In the United States, well over half of all aluminum use is accounted for by the food packaging and transportation industries. For beverage containers, alternative materials such as glass can be used. However, aircraft, automobiles, and bicycles all currently rely heavily on aluminum.19
Much of the world's stock of aluminum, with its light weight and strength, is invested in the fleet of commercial planes. At any given time, a substantial fraction of the world's aluminum is actually airborne. With air travel expanding at 6 percent a year, the investment of aluminum in aircraft is also expanding.20
Although the use of aluminum in aircraft is well established, the substitution of aluminum for steel in automobiles is more recent, spurred by rising fuel prices and the desire for better gasoline mileage. Aluminum use in the average American automobile, for example, climbed from 87 kilograms in 1991 to 110 kilograms by the end of the decade. Although aluminum costs far more than steel, the lower weight of a vehicle with aluminum reduces fuel use, which over the lifetime of a car can more than offset the extra energy used to produce aluminum.21
Aluminum production exacts a heavy environmental toll as well, through both the mining and the smelting processes. Because aluminum typically occurs in thin layers of bauxite ore, extracting it by surface mining scars the landscape. For each ton of aluminum produced, a ton of "red mud"—a caustic brew of chemicals--is left after the bauxite is extracted. This red muck is left untreated in large, biologically lifeless ponds, eventually polluting both surface and underground water supplies.22
But most of the damage done by aluminum production comes from generating electricity to run the smelters. Worldwide, the aluminum industry uses as much electric power as the entire continent of Africa. In some cases, the electricity for aluminum smelting comes from coal-fired power plants, but often it comes from hydroelectricity. Scores of dams have been built, particularly in remote regions, to produce cheap electricity to manufacture aluminum. Governments eager to build indigenous industry in their countries compete with each other for aluminum smelters by subsidizing the cost of electricity. As a result, aluminum is one of the world's most heavily subsidized raw materials.23
Among the metals, gold is distinguished by two things—its minute production and vast environmental disruption. In 1991, producing a meager 2,445 tons of gold required the removal and processing of more than 741 million tons of ore—a mass equal to nearly two thirds of the iron ore used to produce 571 million tons of iron that year. (See Table 6-3.) The leading gold producer is South Africa. Other producers include Australia, Brazil, Russia, and the United States. Eighty-five percent of the gold mined goes into jewelry.24
Beginning in the nineteenth century, gold was used to guarantee the value of paper currencies. As a result, much of the world's gold is stored in the vaults of national banks. Once the United States moved off the gold standard in 1971, however, many countries followed suit, and some have since sold gold from their vaults, including Australia, the Bank of England, the Netherlands, and the Swiss National Bank. This means that gold is being transformed from the final barometer of the value of paper currency to just another commodity. The Economist observes that gold is "the spent fuel of an obsolete monetary system."25
In damage per ton of metal produced, nothing comes close to gold. Each ton of gold requires the processing of roughly 300,000 tons of ore—the equivalent of a small mountain. Over the last decade, a new technique of processing gold ore, called cyanide heap leaching, has come into widespread use. Cyanide solution is leached through a pile of crushed ore, picking up bits of gold as it passes through. This reduces the cost of gold mining, but it leaves behind toxic waste. Cyanide is so toxic that the ingestion of a teaspoon of 2 percent cyanide solution will lead to death within 40 seconds.26
In January 2000, a giant spill of 130 million liters of cyanide solution from a gold mine in Romania drained into the Tisza River, flowed through Hungary into Yugoslavia, merged with the Danube, and emptied into the Black Sea. The lethal solution from the Australian-operated mine left an estimated 1 million kilograms of dead fish in the Hungarian segment of the river alone. This cyanide spill, which left long stretches of river lifeless, has been called Europe's worst environmental disaster since Chernobyl.27
Cyanide spills have occurred in many countries. A similar incident in 1992 in the Alamosa River, a tributary of the Colorado River in the United States, killed everything in a 17-mile stretch and left the state of Colorado with a $170-million cleanup bill after the company responsible declared bankruptcy.28
Another common mining technology uses mercury to extract gold from ore. Mercury accumulates in the environment, concentrating as it moves up the food chain. It was discharges of mercury into Japan's Minamata Bay a generation ago that demonstrated the brain damage and birth defects this heavy metal can cause.29
In the Amazon, gold miners release 200,000 pounds of mercury each year into the ecosystem, reports John Young. Although mercury levels in fish in the Amazon often exceed the levels for safe human consumption, people in the area have no alternative protein source. One teaspoon of mercury in a 25-acre lake can render fish unsafe for human consumption. No one knows when the effects of mercury intake will begin to show up as brain damage and birth defects in the Amazon, but we do know that they first appeared in Japanese infants roughly a decade after fertilizer plants began releasing mercury into Minamata Bay.30
Aside from the discharge of highly toxic cyanide and mercury into the ecosystem, gold mining is also a physically dangerous activity. In South Africa, where most of the gold comes from underground, death in the mines is routine, claiming one life for each ton of gold produced.31
Gold is not the only metal that is damaging the planet. The extraction of other metals, such as copper, lead, and zinc, also disfigures the landscape and pollutes the environment. Reducing this destruction of the natural landscape and the pollution of air, water, and soil depends on designing a new materials economy, one where mining industries are largely replaced by recycling industries.
| Table 6-2. World Production of Nonmetallic Minerals | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Source: See endnote 12. |
| Table 6-3. Metal Production and Ore Mined for Each Metal, 1991 | ||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
| Source: U.S. Geological Survey; John E. Young, Mining the Earth (Washington, DC: Worldwatch Institute, July 1992); W.K. Fletcher, Department of Earth and Ocean Sciences, University of British Columbia |
ENDNOTES:
12. Table 6-2 from USGS, op. cit. note 4, with stone, sand and gravel, and clays from Young, op. cit. note 4.
13. Young, op. cit. note 4, p. 9.
14. Ibid.
15. Soviet Union, United States, and China in IISI, op. cit. note 7; Figure 6-1 compiled from IISI; metals production from USGS, op. cit. note 4.
16. Historical steel production to 1995 from Hal Kane, "Steel Production Rebounds Slightly," in Lester R. Brown et al., Vital Signs 1996 (New York: W.W. Norton & Company, 1996), p. 79; current steel production of 833 million tons from USGS, op. cit. note 4; population from United Nations, op. cit. note 5.
17. USGS, op. cit. note 4.
18. John E. Young, "Aluminum Production Keeps Growing," in Worldwatch Institute, Vital Signs 2001 (New York: W.W. Norton & Company, 2001), p. 64; Australia and other countries from John E. Young, "Aluminum's Real Tab," World Watch, March/April 1992, p. 27.
19. The Aluminum Association, Inc., "Aluminum Facts at a Glance," fact sheet (Washington, DC: June 2000).
20. Lisa Mastny, "World Air Traffic Soaring," in Lester R. Brown et al., Vital Signs 1999 (New York: W.W. Norton & Company, 1999), pp. 86-87.
21. The Aluminum Association, Inc., "Aluminum: An American Industry in Profile" (Washington, DC: 2000), p. 2; Carole Vaporean, "Aluminum Moves to Third Place in Car Content," Reuters, 16 February 2001.
22. Young, "Aluminum's Real Tab," op. cit. note 18.
23. Electricity use by aluminum industry from Young, "Aluminum Production Keeps Growing," op. cit. note 18; African electricity use in 1999 from U.S. Department of Energy, Energy Information Agency, "World Total Net Electricity Consumption, 1990-1999," www.eia.doe.gov/emeu/iea/table62.html; Young, op. cit. note 4, p. 26.
24. Payal Sampat, "Gold Loses Its Luster," in Lester R. Brown et al., Vital Signs 2000 (New York: W.W. Norton & Company, 2000), pp. 80-81; 85 percent of gold in jewelry from "Don't Mine Gold for Jewels," Reuters, 10 December 2000.
25. Sampat, op. cit. note 24; "Central-Bank Gold: Melting Away," The Economist, 4 April 1992.
26. Young, op. cit. note 6, pp. 22-23.
27. "Hungary Seeks Millions in Damages for Cyanide Spill," Associated Press, 11 July 2000; worst since Chernobyl from "International Mining Groups Call for Worldwide Mining Law Reforms," press release (Washington, DC: Friends of the Earth, Mineral Policy Center, and Mineral Policy Institute, 15 December 2000).
28. Timothy Egan, "The Death of a River Looms Over Choice for Interior Post," New York Times, 7 January 2001; "Cyanide-Spill Suit Is Settled in Colorado," New York Times, 24 December 2000.
29. Young, op. cit. note 6, p. 25; Minamata Bay update in Peter Hadfield, "Court Win Follows 40 Years of Suffering," South China Morning Post, 2 May 2001.
30. John E. Young, "Gold Production at Record High," in Lester R. Brown et al., Vital Signs 1994 (New York: W.W. Norton & Company, 1994), pp. 82-83; Patricia Glick, The Toll From Coal (Washington, DC: National Wildlife Federation, April 2000), p. 9.
31. Roger Moody, "The Lure of Gold: How Golden Is the Future?" Panos Media Briefing No. 9 (London: Panos Institute, 1996).
Copyright © 2001 Earth Policy Institute

